| tryptophanase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
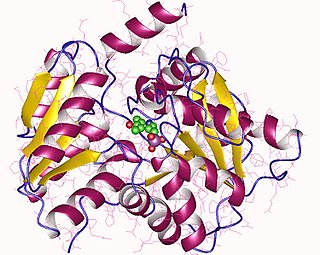
 Tryptophanase tetramer, E.Coli | |||||||||
| Identifiers | |||||||||
| EC no. | 4.1.99.1 | ||||||||
| CAS no. | 9024-00-4 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
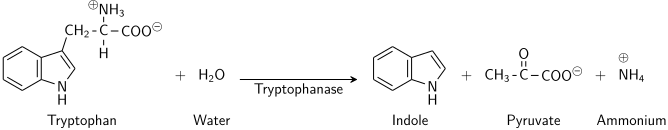
The enzyme tryptophanase (EC 4.1.99.1) catalyzes the chemical reaction
Contents
- L-tryptophan + H2O indole + pyruvate + NH3
This enzyme belongs to the family of lyases, specifically in the "catch-all" class of carbon-carbon lyases. The systematic name of this enzyme class is L-tryptophan indole-lyase (deaminating; pyruvate-forming). Other names in common use include L-tryptophanase, and L-tryptophan indole-lyase (deaminating). This enzyme participates in tryptophan metabolism and nitrogen metabolism. It has 2 cofactors: pyridoxal phosphate, and potassium. [1] [2] [3]