Related Research Articles
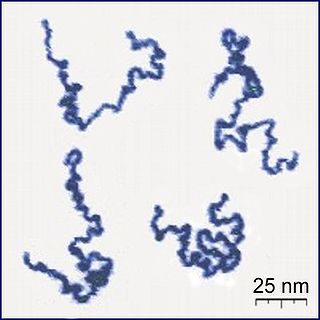
A polymer is a substance or material consisting of very large molecules, or macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic and natural polymers play essential and ubiquitous roles in everyday life. Polymers range from familiar synthetic plastics such as polystyrene to natural biopolymers such as DNA and proteins that are fundamental to biological structure and function. Polymers, both natural and synthetic, are created via polymerization of many small molecules, known as monomers. Their consequently large molecular mass, relative to small molecule compounds, produces unique physical properties including toughness, high elasticity, viscoelasticity, and a tendency to form amorphous and semicrystalline structures rather than crystals.

Molecular engineering is an emerging field of study concerned with the design and testing of molecular properties, behavior and interactions in order to assemble better materials, systems, and processes for specific functions. This approach, in which observable properties of a macroscopic system are influenced by direct alteration of a molecular structure, falls into the broader category of “
Smart materials, also called intelligent or responsive materials, are designed materials that have one or more properties that can be significantly changed in a controlled fashion by external stimuli, such as stress, moisture, electric or magnetic fields, light, temperature, pH, or chemical compounds. Smart materials are the basis of many applications, including sensors and actuators, or artificial muscles, particularly as electroactive polymers (EAPs).

Self-healing materials are artificial or synthetically-created substances that have the built-in ability to automatically repair damages to themselves without any external diagnosis of the problem or human intervention. Generally, materials will degrade over time due to fatigue, environmental conditions, or damage incurred during operation. Cracks and other types of damage on a microscopic level have been shown to change thermal, electrical, and acoustical properties of materials, and the propagation of cracks can lead to eventual failure of the material. In general, cracks are hard to detect at an early stage, and manual intervention is required for periodic inspections and repairs. In contrast, self-healing materials counter degradation through the initiation of a repair mechanism that responds to the micro-damage. Some self-healing materials are classed as smart structures, and can adapt to various environmental conditions according to their sensing and actuation properties.
The term ‘polymer’ refers to large molecules whose structure is composed of multiple repeating units and the prefix ‘supra’ meaning ‘beyond the limits of’. Supramolecular polymers are a new category of polymers that can potentially be used for material applications beyond the limits of conventional polymers. By definition, supramolecular polymers are polymeric arrays of monomeric units that are connected by reversible and highly directional secondary interactions–that is, non-covalent bonds. These non-covalent interactions include van der Waals interaction, hydrogen bonding, π-π stacking, metal coordination, halogen bonding, chalcogen bonding, and host–guest interaction. The direction and strength of the interactions are precisely tuned so that the array of molecules behaves as a polymer in dilute and concentrated solution, as well as in the bulk.
pH sensitive or pH responsive polymers are materials which will respond to the changes in the pH of the surrounding medium by varying their dimensions. Materials may swell, collapse, or change depending on the pH of their environment. This behavior is exhibited due to the presence of certain functional groups in the polymer chain. pH-sensitive materials can be either acidic or basic, responding to either basic or acidic pH values. These polymers can be designed with many different architectures for different applications. Key uses of pH sensitive polymers are controlled drug delivery systems, biomimetics, micromechanical systems, separation processes, and surface functionalization.
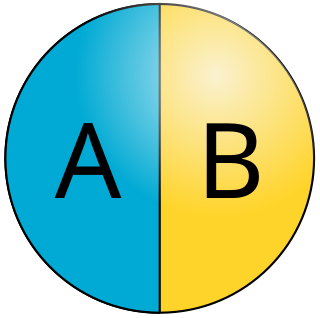
Janus particles are special types of nanoparticles or microparticles whose surfaces have two or more distinct physical properties. This unique surface of Janus particles allows two different types of chemistry to occur on the same particle. The simplest case of a Janus particle is achieved by dividing the particle into two distinct parts, each of them either made of a different material, or bearing different functional groups. For example, a Janus particle may have one-half of its surface composed of hydrophilic groups and the other half hydrophobic groups, the particles might have two surfaces of different color, fluorescence, or magnetic properties. This gives these particles unique properties related to their asymmetric structure and/or functionalization.
Poly(N-isopropylacrylamide) is a temperature-responsive polymer that was first synthesized in the 1950s. It can be synthesized from N-isopropylacrylamide which is commercially available. It is synthesized via free-radical polymerization and is readily functionalized making it useful in a variety of applications.

Temperature-responsive polymers or thermoresponsive polymers are polymers that exhibit a drastic and discontinuous change of their physical properties with temperature. The term is commonly used when the property concerned is solubility in a given solvent, but it may also be used when other properties are affected. Thermoresponsive polymers belong to the class of stimuli-responsive materials, in contrast to temperature-sensitive materials, which change their properties continuously with environmental conditions. In a stricter sense, thermoresponsive polymers display a miscibility gap in their temperature-composition diagram. Depending on whether the miscibility gap is found at high or low temperatures, an upper or lower critical solution temperature exists, respectively.
John Texter is an American engineer, chemist, and educator, and is professor of polymer and coating technology at Eastern Michigan University (EMU) in Ypsilanti, Michigan. He is best known for his work in applied dispersion technology and small particle science, for his international conference organization activities, including Particles 2001, Particles 2002, etc., and the Gordon Research Conferences, Chemistry at Interfaces and Chemistry of Supramolecules and Assemblies, and for his editing of the Primers page for nanoparticles.org.

Takuzo Aida is a polymer chemist known for his work in the fields of supramolecular chemistry, materials chemistry and polymer chemistry. Aida, who is the Deputy Director for the RIKEN Center for Emergent Matter Science (CEMS) and a professor in the Department of Chemistry and Biotechnology, School of Engineering, at the University of Tokyo, has made pioneering contributions to the initiation, fundamental progress, and conceptual expansion of supramolecular polymerization. Aida has also been a leader and advocate for addressing critical environmental issues caused by plastic waste and microplastics in the oceans, soil, and food supply, through the development of dynamic, responsive, healable, reorganizable, and adaptive supramolecular polymers and related soft materials.
Ludwik Leibler, born in 1952 is a Polish-born French physicist. He is Professor of École supérieure de physique et de chimie industrielles de la ville de Paris and member of the French Academy of Sciences and National Academy of Engineering.
Polymeric materials have widespread application due to their versatile characteristics, cost-effectiveness, and highly tailored production. The science of polymer synthesis allows for excellent control over the properties of a bulk polymer sample. However, surface interactions of polymer substrates are an essential area of study in biotechnology, nanotechnology, and in all forms of coating applications. In these cases, the surface characteristics of the polymer and material, and the resulting forces between them largely determine its utility and reliability. In biomedical applications for example, the bodily response to foreign material, and thus biocompatibility, is governed by surface interactions. In addition, surface science is integral part of the formulation, manufacturing, and application of coatings.
The surface chemistry of paper is responsible for many important paper properties, such as gloss, waterproofing, and printability. Many components are used in the paper-making process that affect the surface.
Self-healing hydrogels are a specialized type of polymer hydrogel. A hydrogel is a macromolecular polymer gel constructed of a network of crosslinked polymer chains. Hydrogels are synthesized from hydrophilic monomers by either chain or step growth, along with a functional crosslinker to promote network formation. A net-like structure along with void imperfections enhance the hydrogel's ability to absorb large amounts of water via hydrogen bonding. As a result, hydrogels, self-healing alike, develop characteristic firm yet elastic mechanical properties. Self-healing refers to the spontaneous formation of new bonds when old bonds are broken within a material. The structure of the hydrogel along with electrostatic attraction forces drive new bond formation through reconstructive covalent dangling side chain or non-covalent hydrogen bonding. These flesh-like properties have motivated the research and development of self-healing hydrogels in fields such as reconstructive tissue engineering as scaffolding, as well as use in passive and preventive applications.

Steven Peter Armes is a Professor of polymer chemistry and colloid chemistry at the University of Sheffield.

Smart inorganic polymers (SIPs) are hybrid or fully inorganic polymers with tunable (smart) properties such as stimuli responsive physical properties. While organic polymers are often petrol-based, the backbones of SIPs are made from elements other than carbon which can lessen the burden on scarce non-renewable resources and provide more sustainable alternatives. Common backbones utilized in SIPs include polysiloxanes, polyphosphates, and polyphosphazenes, to name a few.
Nancy Sottos is an American materials scientist and professor of engineering. She is the Donald B. Willet Professor of Engineering and the Head of the Department of Materials Science and Engineering at the University of Illinois at Urbana–Champaign. She is also a co-chair of the Molecular and Electronic Nanostructures Research Theme at the Beckman Institute for Advanced Science and Technology. She heads the Sottos Research Group.
Nanoparticle drug delivery systems are engineered technologies that use nanoparticles for the targeted delivery and controlled release of therapeutic agents. The modern form of a drug delivery system should minimize side-effects and reduce both dosage and dosage frequency. Recently, nanoparticles have aroused attention due to their potential application for effective drug delivery.
Thomas H. Epps, III is an American chemist and the Thomas & Kipp Gutshall Professor of Chemical & Biomolecular Engineering at the University of Delaware. He has a joint appointment in Materials Science & Engineering, and an affiliated appointment in Biomedical Engineering. He serves as the Director of the Center for Research in Soft Matter & Polymers, the Director of the Center for Hybrid, Active, and Responsive Materials, and the Co-Director of the Center for Plastics Innovations. His research considers considers the design, synthesis, characterization, and application of nanostructure-containing polymers related to biobased materials, drug delivery, alternative energy (batteries), nanotemplating, and composite-based personal-protective equipment. He is also the co-founder of Lignolix, which is focused on the valorization of biomass waste.
References
- ↑ Fountain, Henry. "A Polymer Coating That Can Heal Itself Thanks to UV Light". New York Times, 13 March 2009.
- ↑ Jonathan Fahey. "Better Than Watching Paint Dry". Forbes, 12 March 2009.
- ↑ "Coatings that 'self-heal' in sun". BBC NEWS, 12 March 2009.
- ↑ "Self-Healing Coating May Erase Gadget Scratches". National Geographic News, 13 March 2009.
- ↑ "When Hurt, Self-Healing Plastics Turn Red". Discovery News, 26 March 2012.
- ↑ "The microbiological minefield". The Economist, 17 May 2013.
- ↑ "Clemson scientists sweet on self-repair tech". USA TODAY. Retrieved 2018-04-09.
- ↑ Lofton, Lynn. "USM professor develops self-repairing plastic material". Mississippi Business Journal, 19 August 2012.