
Chemokines, or chemotactic cytokines, are a family of small cytokines or signaling proteins secreted by cells that induce directional movement of leukocytes, as well as other cell types, including endothelial and epithelial cells. In addition to playing a major role in the activation of host immune responses, chemokines are important for biological processes, including morphogenesis and wound healing, as well as in the pathogenesis of diseases like cancers.

The innate immune system or nonspecific immune system is one of the two main immunity strategies in vertebrates. The innate immune system is an alternate defense strategy and is the dominant immune system response found in plants, fungi, prokaryotes, and invertebrates.

In the cellular biology, stress granules are biomolecular condensates in the cytosol composed of proteins and RNA that assemble into 0.1–2 μm membraneless organelles when the cell is under stress. The mRNA molecules found in stress granules are stalled translation pre-initiation complexes associated with 40S ribosomal subunits, translation initiation factors, poly(A)+ mRNA and RNA-binding proteins (RBPs). While they are membraneless organelles, stress granules have been proposed to be associated with the endoplasmatic reticulum. There are also nuclear stress granules. This article is about the cytosolic variety.
Extracellular space refers to the part of a multicellular organism outside the cells, usually taken to be outside the plasma membranes, and occupied by fluid. This is distinguished from intracellular space, which is inside the cells.

Molluscum contagiosum virus (MCV) is a species of DNA poxvirus that causes the human skin infection molluscum contagiosum. Molluscum contagiosum affects about 200,000 people a year, about 1% of all diagnosed skin diseases. Diagnosis is based on the size and shape of the skin lesions and can be confirmed with a biopsy, as the virus cannot be routinely cultured. Molluscum contagiosum virus is the only species in the genus Molluscipoxvirus. MCV is a member of the subfamily Chordopoxvirinae of family Poxviridae. Other commonly known viruses that reside in the subfamily Chordopoxvirinae are variola virus and monkeypox virus.

Chemokine ligand 20 (CCL20) or liver activation regulated chemokine (LARC) or Macrophage Inflammatory Protein-3 (MIP3A) is a small cytokine belonging to the CC chemokine family. It is strongly chemotactic for lymphocytes and weakly attracts neutrophils. CCL20 is implicated in the formation and function of mucosal lymphoid tissues via chemoattraction of lymphocytes and dendritic cells towards the epithelial cells surrounding these tissues. CCL20 elicits its effects on its target cells by binding and activating the chemokine receptor CCR6.

David S. Eisenberg is an American biochemist and biophysicist best known for his contributions to structural biology and computational molecular biology. He has been a professor at the University of California, Los Angeles since the early 1970s and was director of the UCLA-DOE Institute for Genomics & Proteomics, as well as a member of the California NanoSystems Institute (CNSI) at UCLA.

Chemokine ligand 21 (CCL21) is a small cytokine belonging to the CC chemokine family. This chemokine is also known as 6Ckine, exodus-2, and secondary lymphoid-tissue chemokine (SLC). CCL21 elicits its effects by binding to a cell surface chemokine receptor known as CCR7. The main function of CCL21 is to guide CCR7 expressing leukocytes to the secondary lymphoid organs, such as lymph nodes and Peyer´s patches.

Chemokine ligand 19 (CCL19) is a protein that in humans is encoded by the CCL19 gene.

Chemokine receptor CXCR3 is a Gαi protein-coupled receptor in the CXC chemokine receptor family. Other names for CXCR3 are G protein-coupled receptor 9 (GPR9) and CD183. There are three isoforms of CXCR3 in humans: CXCR3-A, CXCR3-B and chemokine receptor 3-alternative (CXCR3-alt). CXCR3-A binds to the CXC chemokines CXCL9 (MIG), CXCL10 (IP-10), and CXCL11 (I-TAC) whereas CXCR3-B can also bind to CXCL4 in addition to CXCL9, CXCL10, and CXCL11.
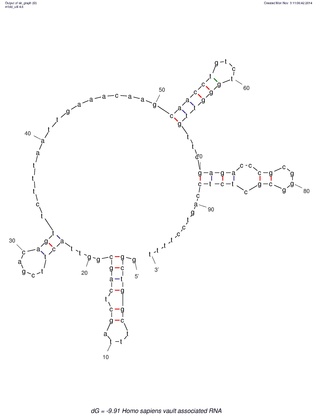
Many eukaryotic cells contain large ribonucleoprotein particles in the cytoplasm known as vaults. The vault complex comprises the major vault protein (MVP), two minor vault proteins, and a variety of small untranslated RNA molecules known as vault RNAs only found in higher eukaryotes. These molecules are transcribed by RNA polymerase III.

Centromere protein B also known as major centromere autoantigen B is an autoantigen protein of the cell nucleus. In humans, centromere protein B is encoded by the CENPB gene.

C-C chemokine receptor type 7 is a protein that in humans is encoded by the CCR7 gene. Two ligands have been identified for this receptor: the chemokines ligand 19 (CCL19/ELC) and ligand 21 (CCL21). The ligands have similar affinity for the receptor, though CCL19 has been shown to induce internalisation of CCR7 and desensitisation of the cell to CCL19/CCL21 signals. CCR7 is a transmembrane protein with 7 transmembrane domains, which is coupled with heterotrimeric G proteins, which transduce the signal downstream through various signalling cascades. The main function of the receptor is to guide immune cells to immune organs by detecting specific chemokines, which these tissues secrete.

C-C chemokine receptor type 11 is a protein that in humans is encoded by the CCRL1 gene.

Major vault protein, also known as lung resistance-related protein is a protein that in humans is encoded by the MVP gene. 78 copies of the protein assemble into the large compartments called vaults.
Mitogen-activated protein kinase 6 is an enzyme that in humans is encoded by the MAPK6 gene.

Fas-activated serine/threonine kinase is an enzyme that in humans is encoded by the FASTK gene.

The vault or vault cytoplasmic ribonucleoprotein is a eukaryotic organelle whose function is not yet fully understood. Discovered and isolated by Nancy Kedersha and Leonard Rome in 1986, vaults are cytoplasmic structures which, when negative-stained and viewed under an electron microscope, resemble the arches of a cathedral's vaulted ceiling, with 39-fold symmetry. They are present in many types of eukaryotic cells and appear to be highly conserved among eukaryotes.
Lymph node stromal cells are essential to the structure and function of the lymph node whose functions include: creating an internal tissue scaffold for the support of hematopoietic cells; the release of small molecule chemical messengers that facilitate interactions between hematopoietic cells; the facilitation of the migration of hematopoietic cells; the presentation of antigens to immune cells at the initiation of the adaptive immune system; and the homeostasis of lymphocyte numbers. Stromal cells originate from multipotent mesenchymal stem cells.
Nancy Kedersha is an American cell biologist and micrographer. She got her Ph.D. from Rutgers University where she worked in Richard Berg's lab studying the characteristics and assembly of prolyl hydroxylases. Afterwards she joined Leonard Rome's lab at UCLA as a post-doctoral fellow where she co-discovered the vault (organelle). Subsequently, she worked at ImmunoGen Inc. where she worked on staining and photographing different cancer cells. She then worked as an instructor of medicine at Brigham and Women's Hospital in Paul Anderson's lab, where her work focused on studying stress granule formation. In late-2020, she retired. In addition to her contributions as a scientist, Kedersha has been quite successful in different microscopy competitions. She is a four-time Nikon Small World finalist and in 2011 she won the Lennart Nilsson Award.















