| Names | |
|---|---|
| Preferred IUPAC name 1H-Indol-3-ol | |
| Identifiers | |
3D model (JSmol) | |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.216.308 |
| EC Number |
|
| KEGG | |
PubChem CID | |
| UNII | |
CompTox Dashboard (EPA) | |
| |
| |
| Properties | |
| C8H7NO | |
| Molar mass | 133.14728 |
| Hazards | |
| GHS labelling: | |
  | |
| Danger | |
| H302, H311, H319, H400 | |
| P264, P270, P273, P280, P301+P312, P302+P352, P305+P351+P338, P312, P322, P330, P337+P313, P361, P363, P391, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
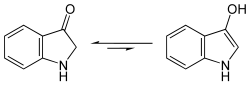
In organic chemistry, indoxyl is a nitrogenous substance with the chemical formula: C8H7NO. [1] [2] Indoxyl is isomeric with oxindol and is obtained as an oily liquid.