| glutamate-1-semialdehyde 2,1-aminomutase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 2epj, Aeropyrum pernix (Archaea) | |||||||||
| Identifiers | |||||||||
| EC no. | 5.4.3.8 | ||||||||
| CAS no. | 68518-07-0 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
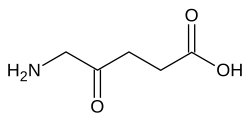
In enzymology, glutamate-1-semialdehyde 2,1-aminomutase (EC 5.4.3.8) is an enzyme that catalyzes the chemical reaction
The enzyme converts its substrate, glutamate-1-semialdehyde into aminolevulinic acid. [1] [2] [3]
This enzyme belongs to the family of isomerases, specifically those intramolecular transferases transferring amino groups. The systematic name of this enzyme class is (S)-4-amino-5-oxopentanoate 4,5-aminomutase. This enzyme is also called glutamate-1-semialdehyde aminotransferase. This enzyme participates in porphyrin and chlorophyll biosynthesis. It employs one cofactor, pyridoxal phosphate. [1]